A speech by a biomedical researcher claiming that 21 percent of patients from the Moderna vaccine trial have experienced severe adverse events has been shared thousands of times on social media since December 11. This claim is false; a report to the US Food and Drug Administration (FDA) shows severe adverse events for just 82 patients, or 0.5 percent of trial participants. Only five cases, or less than 0.1 percent, were linked to the vaccine itself, which medical experts said is within the standard range for vaccine trials.
"Watch this before it is taken down...VERY IMPORTANT," reads a December 11, 2020 Facebook post that includes a video of a speech at the Pennsylvania state Capitol that has been shared more than 18,000 times.
Screenshot of a Facebok post taken on January 11, 2021 The video appears to have been recorded on October 20, 2020, according to captions, and copies of it which have garnered hundreds of thousands of views on YouTube. In it, biomedical researcher James Lyons-Weiler speaks about the purported dangers of Covid-19 vaccines.
Also Read: US Vice President Mike Pence Did Not Fake Getting COVID-19 Vaccine
In his speech, he claims that "the only single data point that we have on what percentage of human patients exposed to coronavirus vaccines have had serious adverse events is from the Moderna trial, and that number is 21 percent. Twenty-one percent of people are having serious adverse events from this vaccine in that trial. The other ones haven't published their data yet."
However, William Moss, Executive Director of the International Vaccine Access Center at the Bloomberg School of Public Health at John Hopkins University, told AFP in an email, "This is false. I do not know where the 21 percent number for serious adverse events come from."
He said the best data on adverse effects was reported to the Vaccines and Related Biological Products Advisory Committee. That data formed "the basis of the Emergency Use Authorizations issued by the Food and Drug Administration (FDA)," he added.
The Moderna Covid-19 vaccine received emergency authorization by the FDA on December 19, 2020. The Pfizer-BioNTech vaccine, which uses the same mRNA technology, had received similar approval a week earlier.
Prior to authorization, Moderna submitted a document to the FDA, the federal agency responsible for approving vaccines, showing that only 82 people, or 0.5 percent of trial patients out of 15,184, experienced adverse effects, with only five, or less than 0.1 percent, linked to the vaccine (see the "Safety" section starting page 31 of the document).
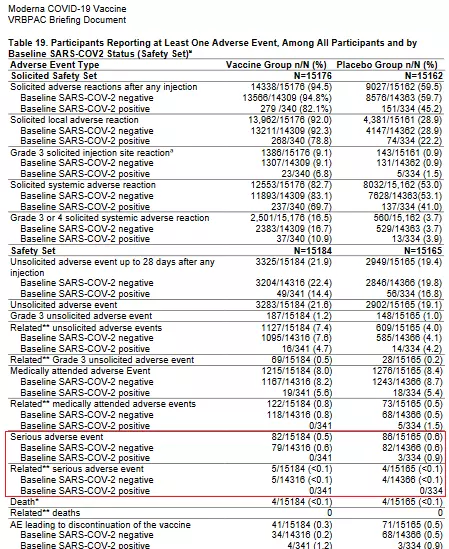
Screenshot of of a document posted on fda.gov, taken on January 11, 2021 Moss explained that "this range in the frequency of serious adverse events is not unexpected" for a vaccine trial.
He also published a blog post clarifying questions around the safety of the Covid-19 vaccines here.
"For both the Pfizer and Moderna vaccines, these reactogenic side effects were mild to moderate, occurred up to two days after vaccination, and do not have any long-term consequences," he wrote.
"Severe adverse reactions – which include any severe disease after vaccination whether related to the vaccine or not – occurred in less than five percent of vaccine recipients," he added.
André Veillette, director of the Molecular Oncology Research Unit at the Montreal Clinical Research Institute (IRCM), also said in an email that the number of reactions was "fairly standard, with adverse events both in vaccine and in placebo groups" including pain, redness, swelling, fever or malaise.
"There are about 15,000 people in each group, so the frequency of adverse events is not high," added Veillette, who joined the Government of Canada Covid-19 vaccine task force in August.
Also Read: No, Patients Did Not Eat Each Other After Getting COVID-19 Vaccine
The US Centers for Diseases Control and Prevention (CDC) published a report about the Pfizer-BioNTech vaccine on January 6, 2020 that concluded that roughly 1 out of 100,000 patients experienced severe allergic reactions.
"As of December 23, 2020, a reported 1,893,360 first doses of Pfizer-BioNTech COVID-19 vaccine had been administered in the United States, and reports of 4,393 (0.2%) adverse events after receipt of Pfizer BioNTech COVID-19 vaccine had been submitted to the Vaccine Adverse Event Reporting System (VAERS)."
These included 21 cases of anaphylaxis, severe allergic reaction, but "No deaths from anaphylaxis were reported after receipt of Pfizer-BioNTech COVID-19 vaccine," the CDC said.
The Moderna vaccine will be the subject of a similar report as more data becomes available, the CDC said.
Contacted by AFP, Moderna did not reply.
AFP Fact Check previously debunked many false or misleading claims about the Covid-19 vaccines here.
(Except for the headline, this story has not been edited by BOOM staff and is published from a syndicated feed.)